All-solid-state-batteries (ASSB)
The future of energy storage
What makes all-solid-state batteries different?
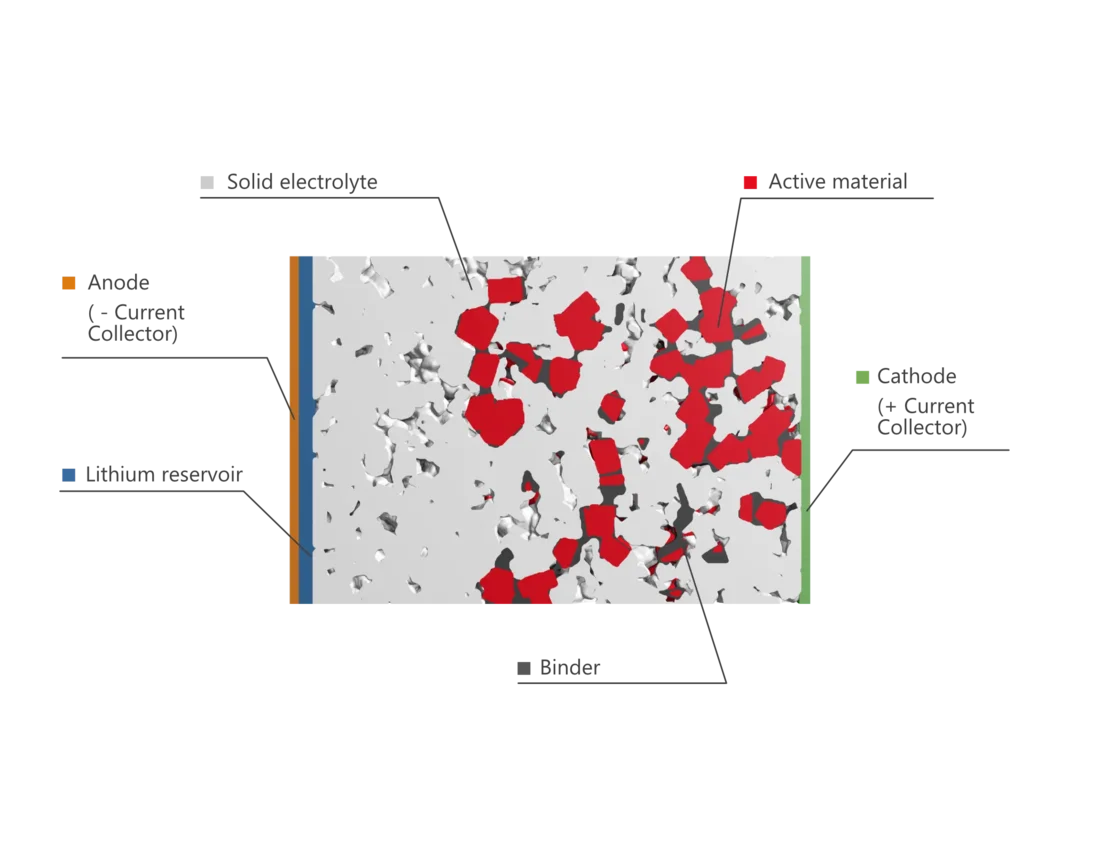
Solid electrolytes are used in all-solid-state batteries (ASSB) instead of the liquid electrolytes of conventional lithium-ion batteries.
All solid-state batteries promise improved safety in everyday use by eliminating the need for liquid electrolytes, which can pose flammability risks in conventional batteries.
The maximum energy density of all-solid-state batteries also exceeds that of conventional batteries. The use of solid electrolytes allows the adoption of metallic lithium anodes, as the solid electrolyte counteracts the risk of dendrite formation and cell shorting.
These characteristics suggest exciting potential for this technology's use in future mobile devices and vehicles.
An all-solid-state battery stores electrical energy by intercalation and de-intercalation of lithium ions in the active materials in the cell. In this process, called (de)lithiation, lithium ions and electrons split and recombine during discharging and charging processes. The lithium ions are transported through the electrolyte to the counter electrode, while electrons are exchanged via the external circuit.
The charging and discharging process:
Step 1: Lithium ions are transferred through the solid electrolyte from the active material of the cathode towards the current collector of the anode. To achieve this, an external voltage supply is connected, to transfer electrons via the cathode current collector to the anode. Lithium ions and electrons recombine at the current collector of the anode to metallic lithium and form a lithium reservoir.
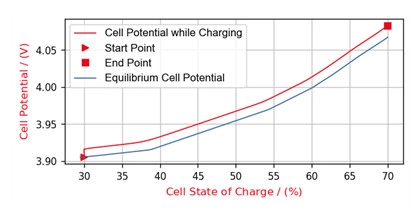
Step 2: During the charging process, the cell voltage may be tracked along the State-Of-Charge of the battery.
Step 3: The discharging process is the reversal from the charging process. The lithium reservoir releases lithium ions that are transported through the electrolyte to the active material in the cathode. The cathode active material gets lithiated again. During this process, electrons pass the external circuit from the anode to the cathode and can power an electrical consumer.
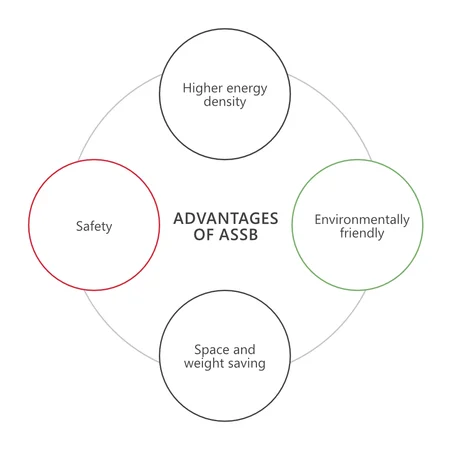
- Higher energy density: By using higher energy materials such as metallic lithium, ASSBs promise higher energy densities of up to 500 Wh/kg combined.
- Temperature resistance and safety: Replacing flammable organic liquid electrolytes with ion-conducting, solid materials significantly reduces the possibility of a fire hazard for all-solid-state-batteries compared to conventional batteries.
- Space and weight saving: New space- and weight-saving cell concepts are possible, such as structural batteries with all-solid-state batteries that enhance mechanical stability.
- Environmental friendliness: All-solid-state batteries are a sustainable alternative to conventional batteries because they do not contain toxic or flammable liquids and are more durable in use. High-capacity ASSBs, such as those used in vehicles, can be used to store excess energy from renewable sources.1
Despite their promise, solid-state batteries are still in the early stages of a development that faces a number of challenges.
The mechanical stress caused by the expansion and contraction of the materials during the charging cycle poses a greater problem for ASSBs than for conventional batteries with liquid electrolyte. Under load, the contacts of the solid electrolyte to the active materials and lithium reservoir may be interrupted. As a result, the ion transfer would no longer be possible and the service life of the batteries would be limited.
Conventionally, experimental research has been used to detect these challenges. However these experiments are cost-, time-, and resource-intensive. The design of new all-solid-state batteries can be significantly improved by simulating the performance and aging processes and their effects on the life cycle of ASSBs, leading to improved designs faster and more efficiently.
The GeoDict software allows the import of 3D scans, with the neccessary segmentation tools, as well as the digital modeling of the microstructure of ASSBs.
AI-based methods for the identification of individual objects and microstructures in imported 3D scans, make it possible to create digital twins for use in parameter studies. GeoDict's built-in material database contains common materials and their physical parameters for assigning real physical specifications during modeling. In addition, the material database is user-editable to add custom material parameters to the database for proprietary use.
In addition to modeling the microstructure of ASSBs, the GeoDict software also performs comprehensive analyses of ASSBs. Numerical methods are used to determine parameters such as tortuosity, conductivity, or diffusivity. Electrochemical charging cycles and the aging processes caused by mechanical stress are simulated easily with GeoDict.
More Info
Video: GeoDict Tutorial - How to model an all-solid-state-battery
The GeoDict software will be extended with advanced modules that take into account the special conditions of ASSBs and make it easier to perform simulations in this field for the digital material design of all-solid-state batteries.
In the DELFIN research project, Math2Market is working with leading research institutes to develop robust methods to simulate charging cycles and aging processes in all-solid-state batteries using GeoDict.
The results of this research are being integrated into the GeoDict software and made available as a simulation tool upon completion of the work phases. GeoDict is thus a valuable contribution to the efficient and resource-saving research and development of modern all-solid-state batteries.